Manuals, specifications, and engineering documentation translated with the precision the content demands.
Technical translation errors have consequences that go beyond meaning. A mistranslated safety instruction in a machine manual can create liability. An error in a pharmaceutical dossier can delay a regulatory approval. An inconsistency in a product specification can cause warranty disputes across markets. We take this seriously.
Technical translation requires a translator who understands the subject matter, not just the language. We maintain a panel of translators with professional backgrounds in engineering, manufacturing, medical devices, pharmaceuticals, software, and industrial processes. Assignment is always by subject matter match, not by language availability.
For manufacturing and engineering documentation, we follow EN ISO 17100 process standards and EN 82079 for operating instructions. These are the standards that matter when documentation is submitted to certifying bodies in German, French, British, or US markets. We do not need to be briefed on these standards â our translators are already familiar.
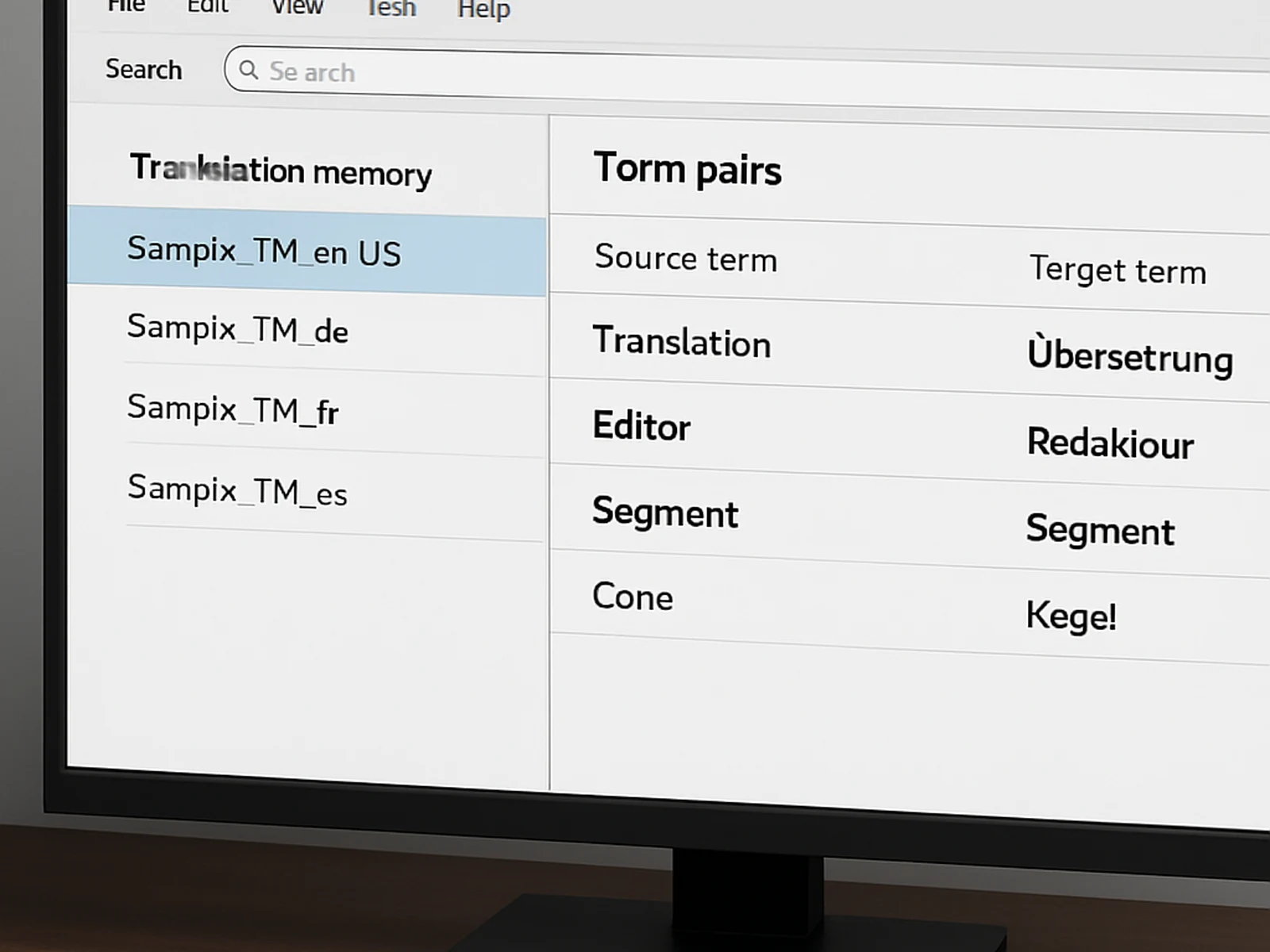
We build a translation memory (TM) and glossary for every client from the first project. Repeated terminology, standard phrases, and approved product names are stored and applied consistently across all future work. This reduces both errors and cost over time. Clients who commission multiple projects benefit directly from this investment in their first project.
Source formats: Word, PDF, Excel, PowerPoint, XML, HTML, InDesign, FrameMaker, MadCap Flare. We return in the same format. For structured data formats (XML, HTML, JSON), we handle the extraction and reinsertion so you receive a ready-to-publish file.
In practice
Common questions
Technical Translation.
Ready to start?
We reply within one business day.